Welcome!
Welcome to the new CancerGRACE.org! Explore our fresh look and improved features—take a quick tour to see what’s new.
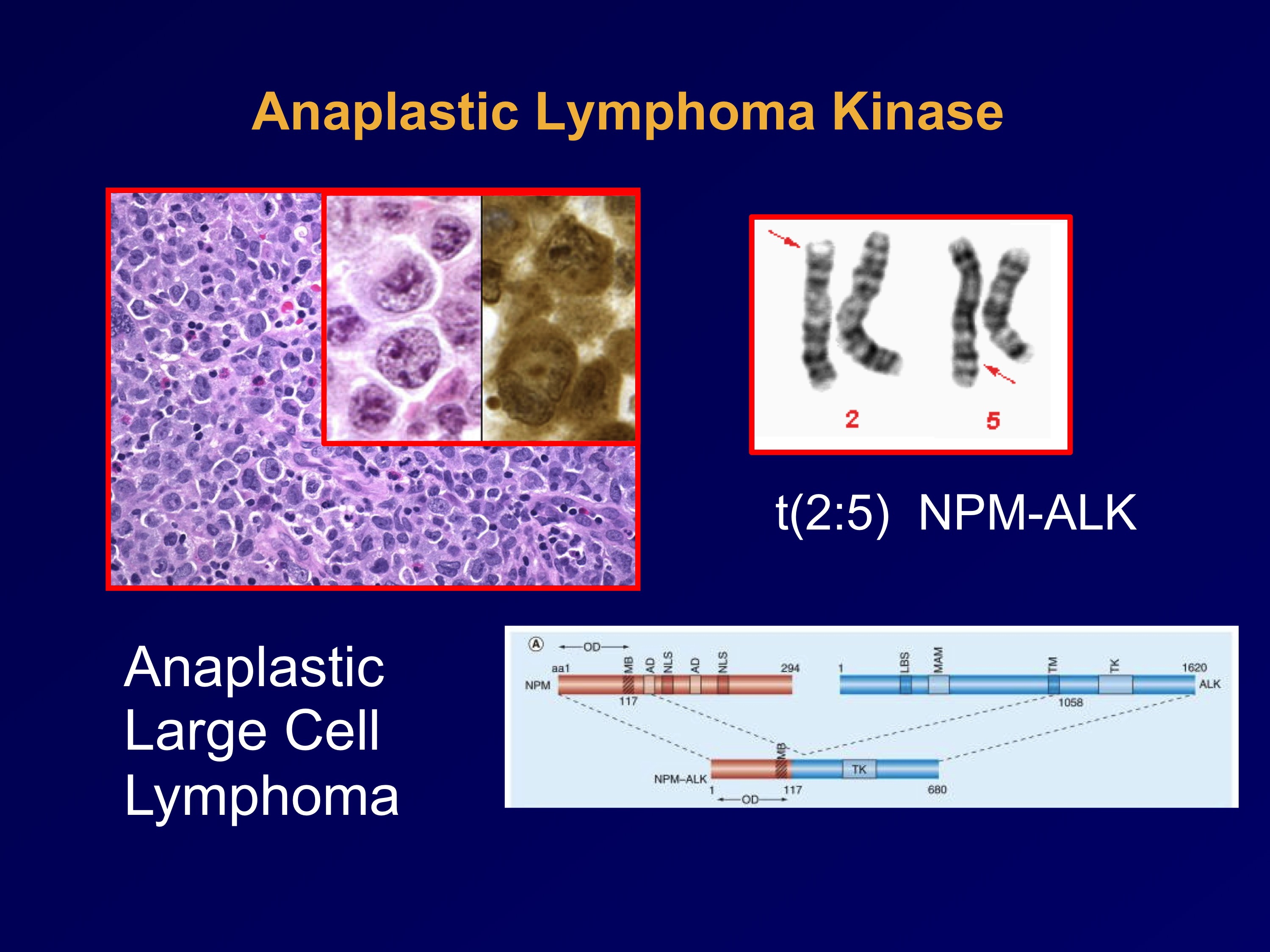
Several weeks ago, we were fortunate enough to be joined by not one but two international stars in lung cancer research that is being translated directly from lab bench to bedside of the patient. I don't think there's a more clear and inspiring example of good science leading to effective therapy, albeit for a limited patient population, than the story of the anaplastic lymphoma kinase (ALK) inhibitor crizotinib (recently FDA approved and commercially launched as XALKORI) for patients with an EML4-ALK rearrangement (approximately 4% of the broader NSCLC population). Drs. Ben Solomon from Peter MacCallum Cancer Centre in Melbourne, Australia, and Ross Camidge from University of Colorado, in Denver, collaborated with a handful of other international researchers from all over the world to study crizotinib and conduct the critical trials, shepherding its development into a treatment now available to help a targeted subset of patients with this targeted therapy.
Dr. Ben Solomon spoke first, providing an overview of the (short) history of the EML4-ALK translocation and how crizotinib began to be studied in the first patients. He then took us on a tour of the highlights of both the efficacy data for this new agent and the side effect profile. Here's the audio version of his presentation:
This is the first of the series of podcasts from this program on ALK inhibition. The next one, coming very soon, will cover Dr. Camidge's presentation, which covered practical issues such as screening for ALK and alternative treatment options for ALK-positive patients. Special thanks to LUNGevity Foundation for partnering with GRACE to make the live program and these subsequent podcasts possible.
Please feel free to offer comments and raise questions in our
discussion forums.
Hi app.92, Welcome to Grace. I'm sorry this is late getting to you. And more sorry your mum is going through this. It's possible this isn't a pancoast tumor even though...
A Brief Tornado. I love the analogy Dr. Antonoff gave us to describe her presentation. I felt it earlier too and am looking forward to going back for deeper dive.
Dr. Singhi's reprise on appropriate treatment, "Right patient, right time, right team".
While Dr. Ryckman described radiation oncology as "the perfect blend of nerd skills and empathy".
I hope any...
My understanding of ADCs is very basic. I plan to study Dr. Rous’ discussion to broaden that understanding.

Here's the webinar on YouTube. It begins with the agenda. Note the link is a playlist, which will be populated with shorts from the webinar on specific topics
An antibody–drug conjugate (ADC) works a bit like a Trojan horse. It has three main components:

Welcome to the new CancerGRACE.org! Explore our fresh look and improved features—take a quick tour to see what’s new.